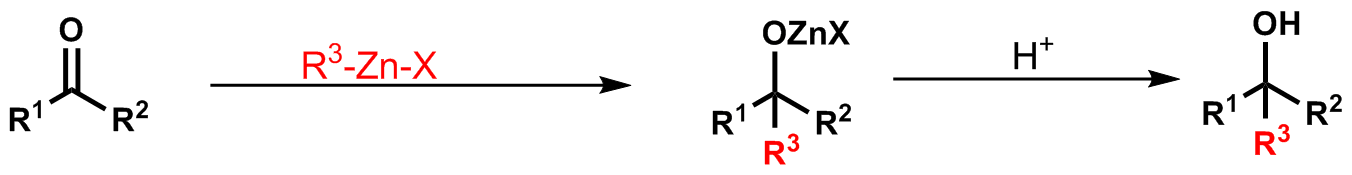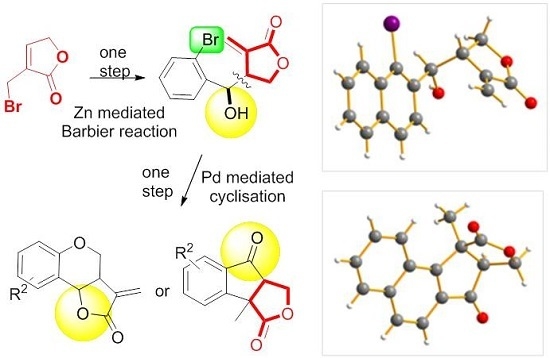
Molecules | Free Full-Text | From α-Bromomethylbutenolide to Fused Tri(Tetra) Cyclic Dihydrofurandiones through Barbier Reaction–Heck Arylation Sequence

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society

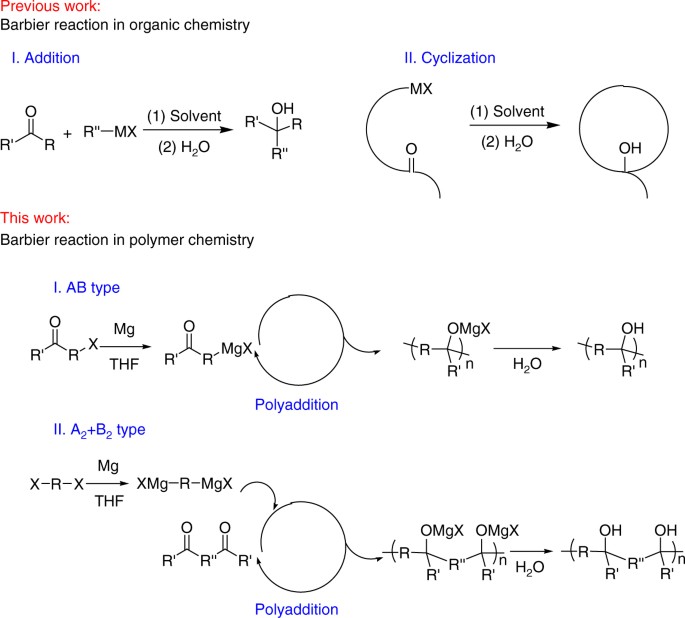
Barbier Reaction Lab Report .docx - Barbier Reaction: An Organometallic Reaction Kiveum Kim Lab Partner: Paul Clark March 15th, 2018 Introduction In | Course Hero

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society

Organozinc compound Barbier reaction Diethylzinc Organic chemistry, organozinc Compound, barbier Reaction png | PNGEgg
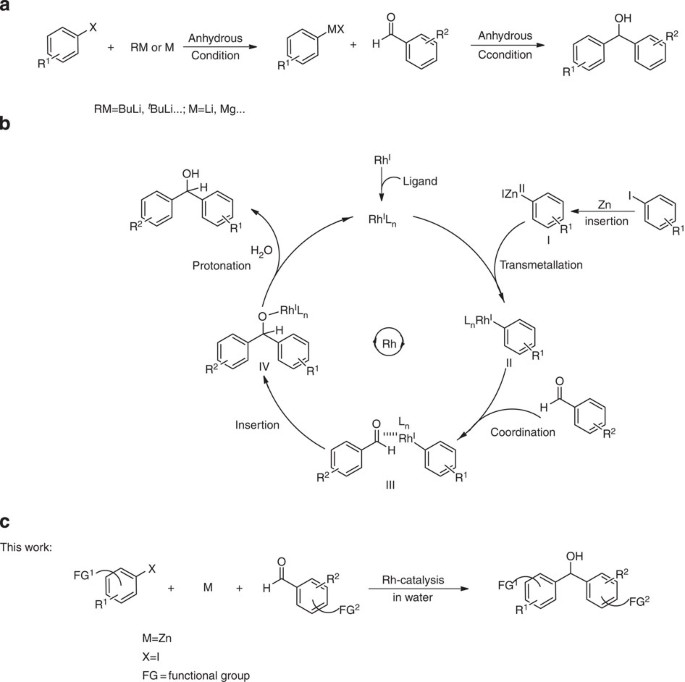
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

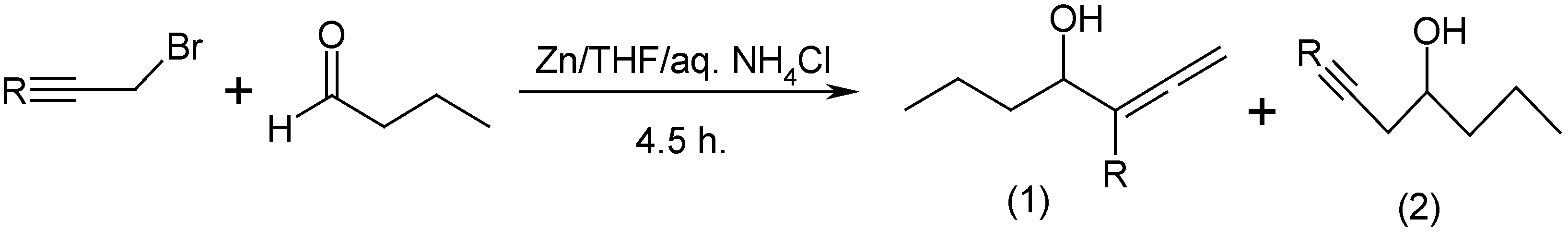
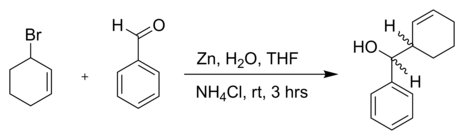
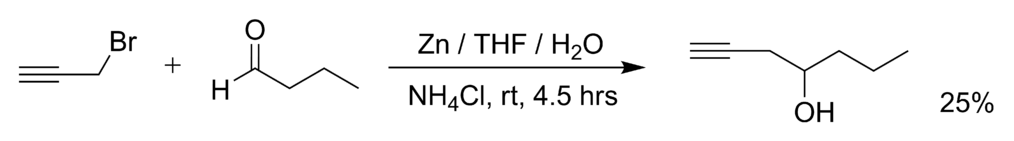
SOLVED: Znls) NH Cl(aq)THF Introduction: The Barbier reaction altemnative t0 thc Grignard reaction between organomagnesium halide and an aldehyde or ketone. Like the Grignard reaction , the product Is either secondary aleohol (
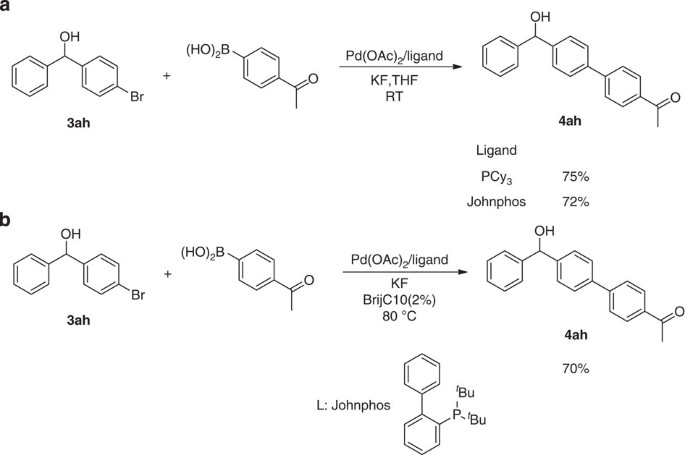
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications